Using flotation reagents correctly means choosing right reagent regime prior to the flotation process. Reagent regime refers to a set of rules regulating the reagent type added, usual quantity fed, and the appropriate ways, position and order of adding reagents.
The reagent regime is mostly determined by factors such as the ore properties, technological process and the required minerals after the ore separability test or semi-industrial test. It is an important element affecting the economic result of ore beneficiation.

Common Types of Reagents
As is mentioned above, the type of reagent used in flotation plant is determined by things like the ore properties, beneficiation process and the required product.
Reagents can be roughly classified into three types according to their effects.
Frothers
Organic surfactant distributed at the air-water interface are used to produce a foam layer that can float minerals. Foaming agents include turpentine, cresol, alcohol, etc.
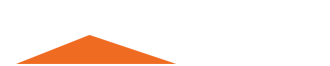
Collectors
They’re used to collect the target mineral. The collectors can change the hydrophobicity of the mineral surface, so that the floating mineral particles adhere to the bubbles. According to their functional properties collectors are divided into three groups, i.e. non-polar, anionic and cationic ones. The commonly used collectors include areoflot, xanthate, diphenylthiourea, fatty acid, fatty amine and mineral oil.

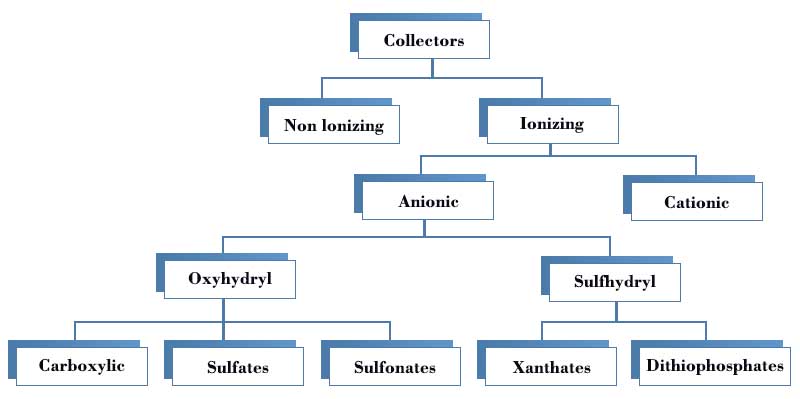
Modifying reagents
Modifying reagents, commonly known as regulators, can change the properties of mineral particles, so as to control the interaction of collectors between individual minerals. With the use of modifying reagents, selective adsorption of collectors on specific minerals can be increased or can be decreased to achieve the separation of individual minerals. Regulators are also used to change the chemical or electrochemical properties of the aqueous medium, for example, to change the pH value and the state of the collector therein.
Modifying reagents can be roughly divided into the following groups.
- pH regulators, e.g. sodium carbonate, sulfuric acid, sulfur dioxide
- Activators, e.g. copper sulfate, sodium sulfide
- Depressants: lime, yellow prussiate of soda, sodium sulfide, sodium sulfur dioxide, cyanide, sodium sulfate, zinc sulfate, potassium dichromate, sodium silicate, tannin, soluble gum, starch, synthetic polymer, etc.
- Other regulators: wetting agent, floatation agent, cosolvent, etc.

Effect of Dosage
The dosage of reagents in flotation should be appropriate. Insufficient or excessive dosage will affect the beneficiation result. Excessive dosage will also increase the cost of beneficiation.
The relationship between the dosage of reagents and flotation result
| Types of reagents | Dosage | Results |
| Frothers | underdose | low recovery rate due to insufficient hydrophobicity of minerals |
| overdose | degrading quality of the concentrate and increased difficulties in mineral separation | |
| Collectors | underdose | poor foam stability |
| overdose | an overflow of the pulp from the froth flotation cell | |
| Activators | underdose | low activating efficiency |
| overdose | inefficient selectivity of minerals | |
| Depressants | underdose | low grade concentrate |
|
overdose |
low recovery rate due to the inhibition of target minerals |
Preparation of Reagents
Dilute the solid reagents into a liquid for easy addition. Reagents with poor water-solubility should be prepared into water solutions with concentration ranging from 2% to 10%. They are, to name a few, xanthate, aerofloat, sodium silicate, sodium carbonate, copper sulfate, sodium sulfide. Insoluble reagents like amine collector should be dissolved in solvents first, and then prepared into aqueous solution. Some such as terpenic oil, aerofloat 31, oleic acid can be directly added. For freely soluble reagents with large quantity to use, the concentration ranges from 10 to 20%. For example, sodium sulfide is normally prepared to a 15% water solution for use in flotation. Reagents that are insoluble in water can be dissolved with the help of an organic solvent, and then prepared into a solution of low concentration.
The preparation of reagents varies with their properties, function, and ways of adding them. The same reagent may differ in the quantity of use and effect due to different ways of preparation.
General ways of preparation are listed as follows.
2-10% water solution
applied for most soluble reagents (e.g. xanthate, copper sulfate, sodium silicate)
non-water solution
applied for some water-insoluble reagents that can dissolve in special solvents (e.g. diphenylthiourea soluble in 10%~20% of aniline solution; aniline aerofloat soluble in alkaline solution of sodium hydroxide)
suspension or emulsion
applied for some insoluble solid reagents (e.g. lime milk in which lime is added into water after being ground into powder, lime can also be directly added to the ball mill and mixing barrel in the form of dry powder)
saponification
applied for most fatty acid collectors (e.g. the saponification of tall oil with the use of oxidized paraffin wax soap in hot 10% sodium carbonate solution in the hematite ore beneficiation process.)
emulsification
Reagents like fatty acids and diesel oil will work better after emulsification by ultrasonic wave or strong mechanical stirring. Many surface active agents can be used as emulsifiers.
acidification
Cationic collectors must be treated with hydrochloric acid or acetic acid because of their poor solubility before they can be dissolved in water for flotation.
aerosol processing
Also known as aerosol flotation, in which the reagents are atomized in the air by using a special spray device and then added into the flotation cell. This method not only improves the floatability of target minerals, but also significantly reduces the dosage of reagents. For example, the dosage of collectors can decrease 2/3 to 3/4, the frothers 4/5.
electrochemical treatment
The electrochemical treatment of reagents in solution by direct current can change the state of the reagents themselves, the pH value and the REDOX potential of the solution, so as to improve the concentration of the most active reagents, the critical concentration at which colloidal particles are formed as well as the degree of dispersion of insoluble reagents in water.
Where to Add Reagents
In order to give full play to the reagents’ work, the general practice of dosing is to add regulators, depressants and part of collectors (such as kerosene) into the ball mill to form a suitable flotation environment as soon as possible.
Collectors and frothers are added in the first flotation mixing tank. The activator should be added into the first tank and the collector and frother the second in case of two tanks.
According to their different roles, reagents are added in different positions. In the case of copper sulfate, xanthate, terpinol oil, the general arrangement is copper sulfate added in the first mixing tank, xanthate added in the second mixing tank, and terpineol oil added at the exit of the second mixing tank.
The flotation plant normally starts with adding pH regulators to modify the pH value of the pulp for collectors and depressants to perform effectively.
Don’t let irrelevant ions ruin your reagents! For example, the reaction of copper ions with the hydride ions deactivates the hydride. Therefore, in the separation process of copper and sulfur, if there are many copper ions in the mixing tank, add cyanide during the flotation operation instead of directly into the mixing tank.
Order of Adding Reagents
The general order of adding reagents in flotation plants is as follows: for the flotation of raw ore, it should be: pH regulators, depressants or activators, frothers and collectors; for the depressed minerals it should be: activators, frothers, collectors.
Ways of Adding Reagents
Flotation reagents can be added simultaneously or separately. Generally, water soluble reagents that are difficult to be taken away by froths and with good stability can be added simultaneously, that is, all reagents to be added at a time prior to the pre-concentration. Otherwise, reagents that are easy to be taken away by froths or react with fine pulp and soluble salts, should be added in different stages.
Recent years a lot of experimental research work has been carried out domestically and abroad in order to improve the efficiency of reagents by using physical methods, including emulsification, heating tanks, aerosol processing, the treatment of electric field and magnetic field, ultraviolet radiation and high energy radiation.